Enzymatic Characterization of Mammalian Cells
Motivation
Mammalian cell cultures are commonly used for the production of recombinant proteins and vaccines. Thus, the knowledge of their metabolic capacity has great potential for process optimization by improving cultivation conditions as well as by designing cell lines using directed genetic modifications. In particular, platforms for the characterization of metabolome, fluxome, and snap shots of a given enzyme activity status/maximum enzyme activities can provide functional and regulatory knowledge about cell metabolism.
Aim of the project
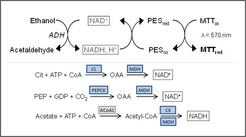
To examine cell metabolism and regulatory interactions in more detail, the project focuses on the measurement of intracellular enzyme activities. Therefore, a high-throughput platform established for enzyme activity measurements in plant cells was first adapted to adherent mammalian cell lines and then further adapted to suspension cell lines (in cooperation with the MPI for Plant Physiology). Changes in enzyme levels are analyzed under different cultivation conditions, primarily focusing on glycolysis, glutaminolysis, citrate cycle, and pentose phosphate pathway. Enzyme activities are essential for the analysis of metabolic and regulatory networks, where disagreements between flux and metabolite levels hint to allosterically controlled enzymes and very low or missing activity levels indicate missing pathways.
For the determination of enzyme activities, cycling assays are used to achieve a very high sensitivity compared to direct enzyme activity measurements. Thus, cell extracts can be monitored at high dilution. Interfering side reactions and concentration of inhibiting substances are also reduced. Furthermore, only very small sample volumes are required, which allow high throughput small-scale cultivations.cale cultivations [1].