Analysis and Mathematical Modelling of IAV DIP Replication
Motivation
Defective interfering particles (DIPs) are composed similarly to influenza A virus (IAV) particles, but they contain an internal deletion in one of their eight viral genome segments. Thus, DIP replication is defective and dependent on co-infection with infectious standard virus (STV). During co-infection, they inhibit replication and packaging of the STV, and induce an antiviral state. Therefore, they are considered as a new class of antiviral drugs.
Aim of the project
As the complex interplay between DIPs and STVs is not yet fully resolved, we investigate the IAV infection dynamics experimentally to establish a novel multiscale model, which describes the DIP/STV replication at the intracellular and extracellular level (Fig. 1). To analyze the time-resolved replication dynamics, we conduct infection experiments for a broad range of STV and DIP concentrations in parallel. For this, a reverse genetics approach was used to generate a helper-virus free segment 1 (S1)-derived DIP seed using a cell line complementing the non-functional polymerase basic protein 2 (PB2) of S1.
The analysis of IAV and DIP replication in a co-infection includes the measurement of extracellular and intracellular viral RNA concentrations by real-time RT-qPCR. Moreover, imaging cytometry is used to provide detailed insights into the status of infection such as the fraction of infected cells and apoptosis. Based on this quantitative and qualitative data, an unprecedented level of detail and resolution is available to validate our mathematical model of DIP replication. This model will support the optimization of cell-culture based DIP production and allow the investigation of critical factors in antiviral therapy, such as the required DIP to STV concentration. Eventually, this model may be further extended to describe the tissue, or even organism level in the future.
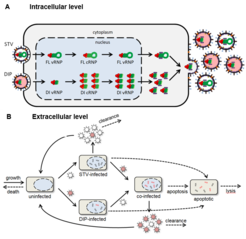
Fig. 1: Influenza A virus replication in the presence of DIPs. A) Intracellular level: Preferential replication of defective interfering (DI) virus ribonucleoproteins (vRNPs) in comparison to full length (FL) vRNPs. The complementary RNA (cRNA) represents the replication intermediate. B) Extracellular level: DIP and STV spreading in a population of cells in a bioreactor (modifed).
